"Is the Inside of an Atom Static?"
("Are Atoms like Planets or Molecules?")
("Do Protons, Neutrons, and Electrons Actually Move Inside Atoms?")
by Ted Huntington
09/24/2004
Note: I am putting this unfinished article on the web because it is a good idea and I want to share what I have. I have not submitted this article for official rejection yet. Because I am opposed to secrecy, and this idea is worthy of exploration. I think this view, that the inside parts of atoms (except for the photons) may be statically held together is on the same plane as the idea that the inside parts of the atom move, because we have yet to see inside the atom, although we see the products (photons) that come out of atoms when they are destroyed or split.
Do protons and neutrons actually move inside atoms? Do electrons orbit atoms or do they stay in one place?
Can you imagine a time when people had never heard of atoms? The history of the human theory of atoms is a wonderful story that most people have never heard. Democritus of Abdera, Greece, is the first human of record to claim that all matter was made of atoms. The word "atom" translates to English as "un-dividable". Because of religious intolerance, brutality and idiocy, not until as recently as 1808 did the secretive and reclusive John Dalton, by experimenting in England with chemistry, move the human species forward by concluding that:
1) all atoms of an element (a distinct matter type, for example Iron, Silver or Gold), have the same size and weight
2) atoms of different elements have different sizes and weights
3) atoms can only combine in whole number ratios (for example 1:1, 1:2,2:3)
4) Atoms can not be destroyed or created (we now know this is not true for atoms, but may be true for photons, particles of light, possibly an even more basic "atom")
The word "element" and "atom" are interchangeable, and "atom" should probably replace "element". Here "element" describes pieces of matter that look the same.
After Dalton, JJ Thompson used the invention of the "cathode ray tube" to beam electrons at sheets of metal. Thompson describes atoms with a model called "the plum pudding model", where electrons are stuck in uniform distribution on a sphere of positive matter. This model was a model of a static atom, an atom where the parts do not move. This model was replaced by Ernest Rutherford, working under JJ Thompson, who explained that atoms have a positively charged nucleus and negatively charged electrons orbiting the nucleus, much in the way planets orbit stars.
Nils Bohr built on this model to describe how light is emitted and absorbed from atoms. Schodinger perfected the model Bohr started, including the model of electron orbits for all the larger atoms.
-do protons, neutrons, electrons constantly move inside atom
-cannot see, may be wrong
-only 2 choices, protons constantly move (current view), protons do not constantly move
-early plum pudding model (not quite structure argument ?)
-planet orbiting model (this branch grew until now)
-in a solid, atoms are stuck together, we can see this with STM
-is valence 8 structure the result of available orbit space, or available structure space?
- orbit space versus structure space
1) orbit like planets
a) electron fits into atom because there is space in outermost orbit (of 8 electrons for example)
b) atom cannot fit electrons because other electrons orbit, no space in valence shell where electrons orbit
c) other atoms connect with this atom (for example Na with Cl, or LiF) because one electron can be shared (either orbiting both atoms, orbiting one atom with timing to make the other atom think an electron is in it's valence shell, or not orbiting, but being stationary between the two atoms)
d) other atoms cannot connect with this atom (for example Na and Ne) because the electrons
2) structure like molecule
a) electrons cannot fit into
exactly 4 protons, neutrons, electrons = true? if yes
-can we model the inside of atoms like we model molecules?
-can the valence shells of 2,8 s,p,d
- it makes sense that as an atom gets larger there is more space for electrons orbiting the nucleus, the same is true for a static atom, there is more surface area for electrons to attach to.
- idea is not that protons, neutrons, electrons are stuck together (although that is also an interesting idea), but that protons, neutrons and electrons can only be a certain distance from each other, leaving a radius of empty space. If some photon group (particle) were to enter that empty space that needs to exist around a proton (for example), the particles would either merge, or be ejected from the atom. In this way an atom can only accept a photon group (for example a proton) that enters perfectly spaced from the rest of the atom. The particle fits in, but does not touch any other particle.
-process of putting together spheres. when two spheres are put together (again sphere represents empty space that must exist around some photon group [particle]), you can see that there is a circle in the center of the peanut like structure, a circle, that represents the closest any new particle can get to those two particles. As the speheres (perhaps of different sizes, depending on the size of the photon group) accumulate, many spaces where a new particle can attach are available, but these spaces may not represent the ideal spacing, which is exactly 1 radius from all particles, so there is some instability introduced. This may help people understand what atoms may be less stable and better for separating (like fission), or what atoms may be more stable, and are better for construction, etc...
everything in the universe is constantly bombarded with photons, sometimes these are single beams of photons, sometimes photons in the form of electrons, protons, neutrons, pions, mu, tau, neutrinos, antiparticles, etc...for an unstable atom, or an atom that has less than ideal spaces between protons and neutrons (which for most atoms and sphere geometries is impossible) these bombarding particles can more easily unseat/dislodge a stationary particle in an atom.
- How good the geometrical fit may determine the stability of the atom. When looking at models of a universe of only 1 particle, the photon, the direction (only, not the velocity) of which is influenced by gravity, although my testing is only in the initial stage, I can see that particle groups tend to collect/bunch/group together. The bigger groups tend to attract most of the photons, it is just like ordering food, most people go to the big food places, voting, most people vote for the main parties, seeing music, most people go to see the most popular music groups, education, most people go to the biggest schools, computers, most people buy from the biggest computer companies, etc... the bigger the photon group the larger the influence the group has on individual photons, and smaller photon groups passing by. One theory of the universe has all the matter eventually collapsing into every space filled with exactly 1 photon/piece of matter, but for billions of years that has not happened, and if space (and perhaps matter) is infinite, there will always be enough space.
But ultimately according to all major theories, matter moves to where there is more matter. These models show that photon groups are balanced by taking on geometrical shapes so that forces between them are balanced. Individual photons easily are sucked into larger photon groups, but when two bigger groups are near each other, the big groups are not as easily influenced, larger groups are not easily sucked into other larger groups. Many times the result is like car manufacturers, there remain 7 or 8 major photon groups in a particular space, each competing for individual photons, but rarely one will get pulled into a larger group (for example, Daimler-Benz buys Chrysler, or HP buys Compaq, for computer companies). So I am thinking of an atom as like a place where 1 to 100 (x2 to include neutrons) car manufacturers are locked in competition.
Ofcourse I may be wrong, and there are complex issues involved in the idea of a static atom (one problem is this: the idea of 8 minimum distance spaces is easy to see with 6 spheres, for example 3 protons and 3 neutrons [a Lithium atom], but when you add 1 more proton and neutron to make Berylium, the geometry is no longer a geometry of 8 minimum spaces, but yet the 8 space valence structure exists for this entire second row of atoms. Possible answer: There may exist a variety of structures in an atom, not just one. A geometry with 8 minimum spaces may exist with the 6 sphere [3 neutron and 3 proton] structure, and other structure may exist next to that structure within a single atom.) This model still should be fully explored and modeled in 3D on computers to see where minimum points (those points where a particle is as close to as many other particles as possible, but no more than 1 radius [which may be different for a proton and neutron] away) exist.
[other possible answer:
keep building up a structure of spheres, and see if 2,8,8,20,20,32,32 patterns emerge, there is some issue about h2 and he2 being diatomic or different atoms]
- distribution of matter, an example is the way a bacteria can be as small as x and as large as .6mm, no smaller or larger bacteria have even been found...any larger than the largest or smaller than the smallest, and perhaps the mechanics of the cell do not function, or something has stopped that from happening, for larger bodies, a different configuration has evolved/succeeded, no human is as large as a planet, at that scale, matter has taken on a different form, no star is as big as a galaxy, at that scale matter takes on a distributed form, presumably different from the inside of a star (or we would have spiral stars). The collective movement and spacing of the trillions and trillions of particles of light makes for a variety of structures (like atoms, bacteria, people, stars and galaxies)
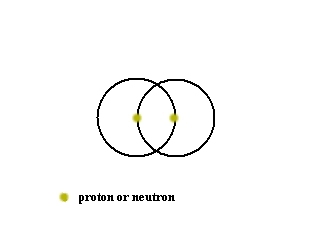
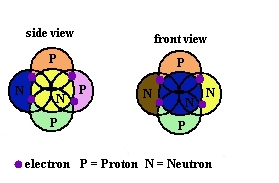

Without being able to actually see inside the atom with the current scanning tunneling microscopes, what experiments can people think of to prove if protons, neutrons and electrons actually are free moving within an atom or statically held in place inside all atoms?
If the inside of atoms moved, wouldn't atoms split in different ways? Do people observe that atoms always split into the same smaller atoms? This is not proof of a static atom, because perhaps the geometry of the hypothetical movement of particles inside of the atom and the nature of splitting atoms with neutrons, for example, makes the split always the same, maybe because protons are always in the same place of their orbit with the neutrons. I am guessing that the current model has neutrons and protons orbiting each other somehow differently than the way electrons orbit the nucleus, similar, perhaps to the way the stars in a multi-star systems move.
In the periodic table I see that the valence/outermost electron layer appears to be ordered like this:
2
8
8
18
18
32
32
Perhaps this is determined by a static shape atoms take. Bohr and Schr�dinger imagined electrons in orbits around a nucleus, I can imagine that, and I want to add the image that maybe the electrons are not orbiting but are filling in spaces in a sphere-like shape.
update 11/16/05:
I have done more experimenting with the idea of static atoms, and made at least one major conclusion: That, if atoms were spherical, electron shells of 2 8 8 18 18 32 32 would not imply that. In addition, what is the real deal with the difference in atomic mass and proton+neutron count? I doubt seriously it is "energy", more likely a phenomenon of the measuring aparatus, or the fact that there are additional particles (in the form of matter) in the atoms. I opened the idea that perhaps there is a dual-nature to atoms that explain the unsphere-like size progression of 2 (8 8) (18 18) (32 32). Presumably the last electron shell is 32. I concluded that there is almost no info on theories of the structure of nucleus shells, only electron shells. The current view claims that all the properties of atoms are determined by the electron shells alone. One experiment I thought of is trying to join atoms without electrons. When Hydrogen and Oxygen are stripped of electrons (if possible), do they still form H2O molecules? If they do, how could they without electrons? I think that would be strong evidence that molecular structure is determine more by the proton and neutron shell structure than by electrons shell structure. If they don't then perhaps that is a useful idea, here we have inert atoms that can be mixed without forming molecules. There is still the basic question of: what is the nature of the electron bond? Do electrons simply fill the space or physically join the other bonded atom? In modeling these atoms, I see that analyzing the structure is complicated. Many times there is a clear shell (as is the case for 13 spheres), but as the structure grows, it is more difficult to determine where clear shells exist (perhaps you can recognize this by looking at the videos below).
Static Atom Models:
----start all current comments on experiments to explore a static atom model--------
conclusions about atom modeling with theory that particle groups are:
1) same size
2) are no closer than a speherical radius of the same size for all groups
3) get as close to all particles as possible/ or only particles that fit in the best position will stick to the atom
main issues:
1) atom is sphere? if yes, problem with (electron and nuclear) shells
2) dual nature of atoms?
3) atomic masses correct? more protons+neutrons than thought, or some other phenomenon?
4) find shells using idea of photons groups that require a certain radius of empty space
5) does the nucleus have shells (like the electrons) too? Does the structure of the nucleus of an atom have anything to do with the way atoms bond, or are the outer-most electrons the only component involved in atom to atom bonding and the properties of all atoms?
1) I found that there are, in some models, for example radius=1, clear "shells", where new photon groups must take a significantly more distant location. But for radii of 2,3,4, and 5 the shells, if any exist were not clearly identifiable.
2) There may be a "dual" nature to atoms on the periodic table. A sphere volume grows exponentially with the equation 4/3*pi*R^3, so if atoms are spherical, their shells should grow in size and number of particles. It is a simple conclusion that as a sphere grows, the amount of surface area (A=4*pi*R) also increases. But, we see the pattern of atoms on the periodic table as increasing, not linearly but in a "double" way. The order of shells between noble gas atoms is represented by particles (protons + neutrons) in groups of: 4,+8,+8,+18,+18,+32,+32 (it is presumed that +32 is this outer shell...the tantalizing atom 118, the inert/noble gas was claimed to have been created in 1995 at Berkeley, but was later retracted...so far no Uuo118 has ever been created publically). So, will this atom be a last shell atom (if it can ever be made)? Will it display properties of an inert gas? So back to the "dual nature" that may exist in atoms. I find it highly doubtful, but have to entertain the possibility that atoms may be built in two parts, atoms may have two basic particles, or spheres of matter. Then, the shells would grow as we would expect spheres to grow: 2+2 8+8 18+18 36+36. There is something beautiful about that model, and it fits the periodic table, but I can't help but think how unlikely it would be for only 1 side of an atom half to fill, while the other half waits...there would undoubtably be combinations of atoms that had, for example, 2+2 3+4 and did not fill: 2+2 0+7. One way, out of the problem that atoms do not grow with a spherical shape is described in more detail
in conclusion
3) that the measurements of atomic mass are correct, but that the interpretation of how many protons and neutrons are in the atoms (with more than 2x mass of protons and neutrons) is incorrect, and that there are actually more protons and neutrons in those atoms. There is something lingering in my mind about how a sphere of spheres grows, and in my experiments, I have found that the spheres do always grow in radius, but...perhaps somehow the way when a sphere grows, the increase of volume creates more spaces to connect on and so ...maybe there is some way that a sphere would have shells of 4 8 and then 8....I have to flush this out and see if there is anything there...I don't think there is.
4) There is an issue about atomic mass. Why is there a difference between the measure atomic mass and the theorized number of protons and neutrons? First the "official" explanation: the extra matter is in "binding energy". I have to doubt that because, I interpret the idea of energy and mass being equivalent, first I doubt the existence of "energy", but I interpret the so-called "energy" as being in a material form, i.e. photons, when in mass form. There are two main possiblities I can think of:
a) the measurements of mass with a mass spectrometer is not accurate, because of some inherent flaw...particles from the spectrometer interfere...heavier atoms have extra problems in a beam that lighter atoms do not have...I am only guessing, there could be a million reasons why.
b) That mass is in the form of neutrons and protons, and there actually are more protons and neutrons in those atoms (and every atom after them). This would mean that Argon, the inert gas, is the first atom that appears to have an extra proton and neutron, for example. This theory tends to help the idea that atoms are spheres. And that is a basic idea. Are atoms spheres or some other shape? If they are spheres, then there is aproblem with the current interpretation. But in addition, there is the major split of two branches of theory that have atoms as rotating/moving versus static (although the photons that compose the protons and neutrons are rotating each other). I don't know which is more accurate, but the rotating atom (although I am the first that I have seen to verbally describe the so-called "nucleus" particles as rotating...I have seen this representation in videos. In other words, people are not proudly and openly proclaiming that protons and neutrons orbit each other to my knowledge, but then only 30% actually believe evolution, so perhaps that is the nature of this and many subsequent centuries).
5) in my experiements, many atoms take on similar shapes...one or more spheres in the center, a row of spheres in circumference (many times 5 or 6), and a group of spheres (many times 2 or 3 spheres) on top and bottom. This is one easier way of looking at these models...how many spheres in: 1) center 2) perimeter 3) top 4) bottom.
6) If integer average distances indicate a new shell (which is doubtful, but maybe there is a similarity and there is insight from presuming such a thing), then the order of spheres (protons+neutrons) in each shell would be something like: 19,63,153, but the atoms on the periodic chart show shells (although they are presumed to be electron shells, the number of protons and neutrons is also equal to the number of electrons) more like 20,39,83 (by atomic mass, or 20,36,72 if going by atomic number) a much more compact structure. So perhaps protons and neutrons form spheres of different sizes. One thing that appears clear is that neutrons are protons+electrons+anti-nutrinos and so perhaps that structure should be modeled.
notes:
1) so-called "alpha particles" or helium nuclei appear to be the only way atoms decay (atoms can be separated in a variety of ways...but people never see/measure naked protons or neutrons? or do they=have to check), so perhaps helium units of 4 particles. If the number of protons and neutrons is correct (the excessive atomic masses are incorrect), then it would tend to support the idea that atoms are built on units of 4. Each inert gas is divisible by 4 (4,20,36,72,108,172,236) can all be made by (1,5,9,18,27,43,59) units of 4. Explore more what kinds of particles are emited from atoms. I know: 1) photons,
2) electrons, 3) helium nuclei, 4) protons?, 5) neutrons? I know that atoms can be split, so maybe...any cmobination of protons, neutrons, etc is possible. After all, burning propane is completely destroying those atoms into photons. Does burning of gases ever result in protons or neutrons?
2) still can check other
a) shapes (ellipsoids, ...)
b) mixed sized shapes (1 larger sphere and 1 smaller sphere)
this kind of testing I think would need to be automated...it would take a large amount of time
3) glue marbles together
4) I think there is a basic idea of electron shells and nucleus (or nuclear) shells. This prompts the experiment of combining two atoms denuded/stripped of electrons...do they still form molecules? or are the electrons necessary components of all atom to atom bonds? Possible examples, include H+ and O+12, do they still form H2O? or NA+11 and CL+17=do they still form NaCl? If no, then is removing electrons a form of making atoms inert from other atoms that do not have electrons? I suppose electrons could be easily removed from neighboring atoms, and it would be difficult to keep atoms without electrons. I just find it hard to believe that nucleus structure has nothing to do with atomic bonding, and that only the electrons connect atoms, that everything depends on the electron shells. Again, it is not clear to me how electrons hold atoms together. Do they orbit both atoms? Do they orbit other electrons from the other atom? Do they temporarily fill a space where an electrons should be in a different atom? A structural model of the atom where electrons are stationary and attached to the other atom is easier to explain and understand.
5) One thing that was kind of a cool thought was that, perhaps somehow, electromagnetic collective gravitational effect (force) can be explained as a difference in size between particles. Only a certain size particle can move through the atomic lattice (mercury as a liquid conducts electricity so that needs to be thought about too). I don't know, but ultimately, I think that electricity is the pulling of matter to a place where there is more matter, basically gravity, but because of some collective group effect, the force is stronger (perhaps multiplied by quantity of particles, or close interactions [like a small ship with Jupiter] gravitational .
questions:
1) shouldn't the atom configurations be the same for all radii? Because the radius of all atoms is the same size. I think the reason there are differences is strictly from integer only differences that would not exist if floating point was used. It seems clear to me that the universe does not function like floating point numbers do, because a particle occupies 1 cubic unit of space, not .5 of a unit of space, etc. It is only the vast difference in size that makes objects appear to have fractional locations, velocities, etc.
2) What are the real differences between using spheres with radius 1, 2, etc. Why do differences exist? What is the representation that should be thought of? Is 1 pixel, 1 of the smallest units of space, or is 1 cubic pixel the smallest unit of space a photon group can fit in?
videos:
This is kind of cool. These are 12 transparent spheres stuck together as closely as possible, but within 1 radius of each other:
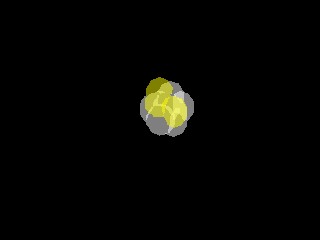 atoms12_trans.avi
atoms12_trans.avi
Here you can see how one more sphere makes 13 spheres which forms a closed shell. Any new sphere will be significantly more distant from the center, in a new outer shell.
 atoms13_trans.avi
atoms13_trans.avi
Here is the 13 sphere shape, more realistically showing how photon groups might gravitationally balance each other. The distances are the same as the video above (10 pixels), but the spheres are only 1 pixel in radius.
 atoms_r1_r10_13.avi
atoms_r1_r10_13.avi
The next shell in the radius=1 pixel model was easy enough to find. It is a 3x3x3 cube:
atoms_r1_27.avi
Again, these shapes evolve naturally, given the formula: for each new sphere (of equal radius) added, find the smallest distance to all spheres, such that this distance is at least as large as the sphere radius. When I did spheres of radius=1, but the minimum distance between their centers could be 10, it was different from the above radius of 1 (I thought it would be the same since radius of 10 is the same as 1 (only everything is *10) in integer only math)
 atoms_r1_r10_27.avi
13 and 27 do not fit the periodic table (4,20,40 does...or perhaps 4,20,36), so this shape is probably not how protons and neutrons are put together. 4x4x4 (64 spheres) doesn't form a cube, because the sphere shape starts to show itself beyond 3x3x3:
atoms_r1_r10_27.avi
13 and 27 do not fit the periodic table (4,20,40 does...or perhaps 4,20,36), so this shape is probably not how protons and neutrons are put together. 4x4x4 (64 spheres) doesn't form a cube, because the sphere shape starts to show itself beyond 3x3x3:
After 13 spheres, finding the next shell was very difficult. I would need to spend more time adding one sphere at a time...sometimes there are consistent geometries, where spheres will be well balanced together (the 13 spheres video above is a good example, you can see 3 on top, 3 on bottom, 6 around the perimeter, and 1 in the center...very nicely balanced), but more often, there is no symmetrical geometry or a very complicated one.
Here are 134 spheres (protons+neutrons) connected one at a time:
 atoms_r1_134b3.avi
atoms_r1_134b3.avi
Here are 200 spheres connected one at a time, colored by distance.
average distance from all other spheres:
1 pixel=red (19)
2 pixels=orange (44)
3 pixels=yellow (109)
4 pixels=green
This model is growing larger faster than the atoms on the periodic table do, so perhaps the spheres are smaller (or in integer representation fit more closely at a size of 8 or 9 pixels, since this should be identical to spheres of .8 and .9). 200 spheres is the equivalent of a Platinum, Gold, Mercury, etc atoms, massive atoms. It would be awesome if we some time could actually build atoms like this.
 atoms_r1_200.avi
atoms_r1_200.avi
When the required minimum distance is 2*radius, the model is like holding a group of marbles together in your hand or with glue. Although, I doubt this model represents the way protons and neutrons hold together.
 atoms_r1_200b.avi
atoms_r1_200b.avi
----this concludes all current comments on modeling static atoms--------



 atoms12_trans.avi
atoms12_trans.avi atoms13_trans.avi
atoms13_trans.avi atoms_r1_r10_13.avi
atoms_r1_r10_13.avi atoms_r1_r10_27.avi
13 and 27 do not fit the periodic table (4,20,40 does...or perhaps 4,20,36), so this shape is probably not how protons and neutrons are put together. 4x4x4 (64 spheres) doesn't form a cube, because the sphere shape starts to show itself beyond 3x3x3:
atoms_r1_r10_27.avi
13 and 27 do not fit the periodic table (4,20,40 does...or perhaps 4,20,36), so this shape is probably not how protons and neutrons are put together. 4x4x4 (64 spheres) doesn't form a cube, because the sphere shape starts to show itself beyond 3x3x3:
 atoms_r1_134b3.avi
atoms_r1_134b3.avi
 atoms_r1_200.avi
atoms_r1_200.avi atoms_r1_200b.avi
atoms_r1_200b.avi